Pressures at the bottom of tanks depend only on the height of the stored water. This has allowed professions using fluids to define meaningful units of pressure expressed in height (or depth) of liquids.
Hence, in our profession of HVAC, we sometimes use the
metre of water [mH2O] which corresponds to the pressure created by such a height of water.
By chance:
1 [bar] ≈ 10 [mH2O]

Question
Knowing that 10 [mH2O] (US: 32.8ft H2O) creates a pressure of 1 [bar], to how many [kPa] does the [mH2O] correspond ?
10 [kPa]
Explanation:
10 [mH2O] (US: 32.8 ft H2O) = 1 [bar]
= 100,000 [Pa] = 100 [kPa]
1 [mH2O] (US: 3.28 ft H2O) = 10 [kPa]
An industrialist working solely with fuel can use the “metre of fuel”.
Fuel being lighter than water, 1[mF] is worth less than the [mH2O].
1 [mF] = 3.28 US ft F = 0.8 [bar]
Seamen and weathermen use the millimetre of mercury [mmHg] [US: in Hg], the unit of pressure corresponding to the height of mercury.
Mercury is much heavier than water:
1 [mmHg] ≈ 133 [Pa] while 1 [mmH2O] ≈ 10 [Pa]
or for example
1 [in Hg] = 3,386 [Pa] while 1 in [H2O] = 249 [Pa]

Question
A diver is at a depth of 15 [m] (US: 49.2 ft).
What pressure does he feel in [bar], in Pascal and in [kPa] ?
150 [kPa]
Explanation:
15 [mH2O] (49.2 ft H2O) = 1.5 [bar]
= 150,000 [Pa] = 150 [kPa]
In our sector, pressures are measured with manometers.
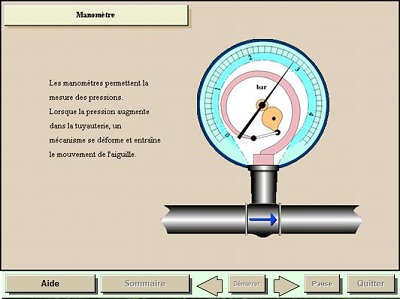
When disconnected, the manometer shows a zero reading. In reality, it should show an ever-present pressure: atmospheric pressure which is approx. 1 [bar].
This pressure is due to the weight of the air pressing on the earth’s surface.
This is what barometers show.

But what interests us is to know if the pressures in our heating or air-conditioning systems are higher or lower than the surrounding air pressure.
- A pressure higher than atmospheric pressure (displayed as > 0 on our manometer) shows us that the system is “under pressure” (pressurized).
- A zero pressure reading shows us that the system is not filled.
- A negative reading (less than zero, showing the system is in “depression”). This could be due to air penetration, not desired as a general rule.
Our manometers, unlike barometers, show the difference in pressure of our systems with regard to atmospheric pressure (either higher or lower). We say that they show a relative or gauge pressure.
Question
What pressure is shown on a manometer installed at the base of a water-tour, 30 meters (98.4 ft) high, in [bar], in Pascal and in [kPa] ?
30 [mH2O] (98.4 ftH2O)
= 3 [bar] = 300,000 [Pa] = 300 [kPa]
Explanation:
The manometer only shows the pressure due to the height of water.
In reality the pressure is really 4 [bar], but what interests us is to know the increase in pressure due to the presence of 30 meters (98.4 ft) of water, so 3 [bar].
